Industrial Bioengineering bridges engineering and biological-medical fields to design, develop, and produce diagnostic and therapeutic devices. This interdisciplinary approach is crucial for revolutionizing human health by creating efficient, customized, and widely available medical solutions. The field demands professionals who can translate research into industrial applications, possessing the engineering expertise to create devices that interact seamlessly with biological systems.
The Master’s Degree in Industrial Bioengineering addresses this need by training professionals with a blend of engineering and medical-biological knowledge. Graduates utilize engineering tools to provide technical and scientific solutions in areas like regenerative medicine, nanomedicine, and personalized therapy. This includes designing medical devices that interact directly with living systems, from biomolecules and cells for diagnostics and therapy to tissues and organs for repair and regeneration.
The curriculum provides fundamental knowledge in biochemistry, biology, medical device design and development, and biomedical data analysis. Students can specialize in diagnostics and modeling, incorporating machine learning, or in science and technology for implantable devices. Elective courses cover biomechanics, neuroengineering, and rapid prototyping.
Industrial Bioengineering graduates are well-equipped for high-level roles in bioengineering (diagnostic and therapeutic devices), biomedical (prosthetics, clinical data analysis), and pharmaceutical (nanomedicine, controlled release) companies. They also find opportunities in wellbeing sectors like cosmetics and nutraceuticals. Bioengineers are key figures in driving innovation, translating laboratory discoveries into clinical practice and market-ready biomedical devices, and facilitating the shift from hospital to personal care.
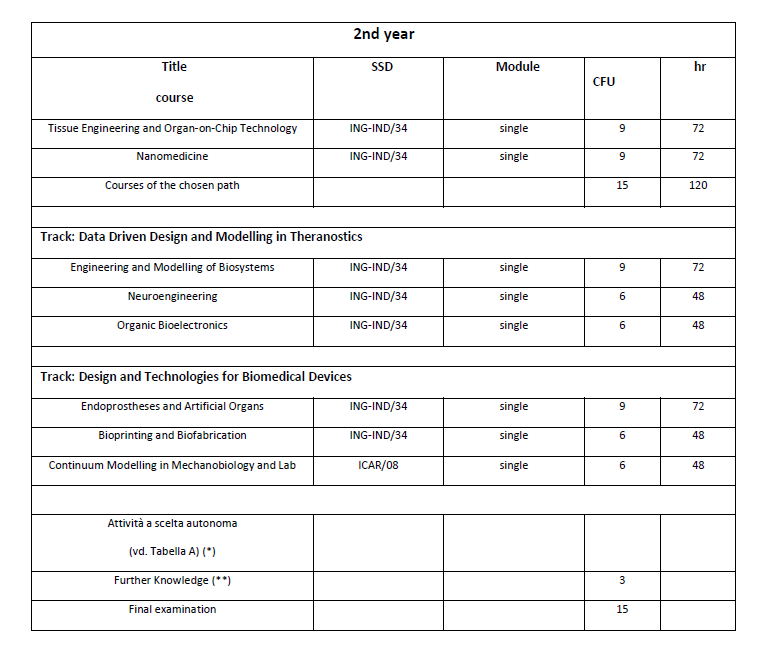
Study Plan
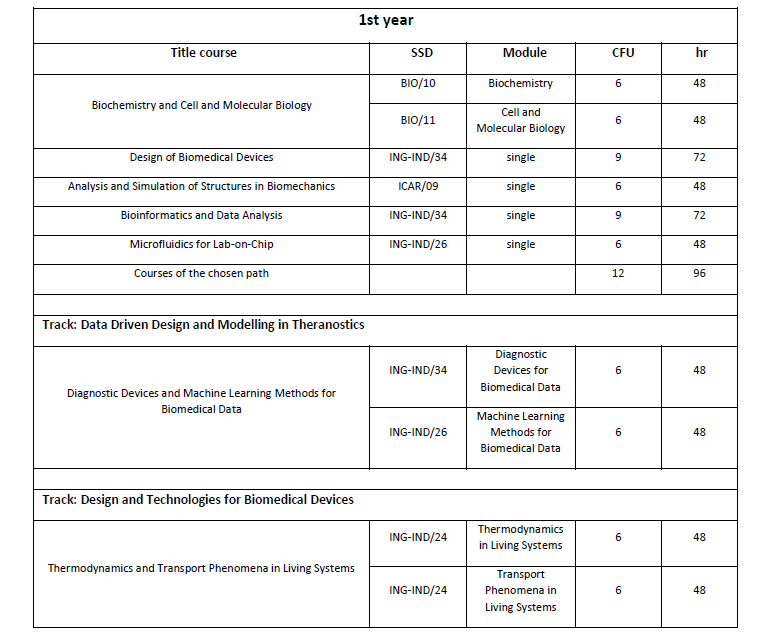
(*) For the choice of the elective courses, the student is free to choose from all the courses activated at the University, or from the courses regulated within the framework of national or international agreements, provided that they are consistent with the training project [RDA Art. 13 c. 4]. The consistency is assessed with an unquestionable judgment by the CCD or by a commission delegated by it. The courses present in Table A are automatically approved and therefore not subject to the evaluation of the CCD. Students are however required to submit the Study Plan form, present on the Degree Course website, within the timeframes established by the CCD. All the courses of the Minor in Applied Machine Learning taught in English are also considered to be automatically applicable (see informatica.dieti.unina.it/index.php/it/laurea-magistrale/minor-in-applied-machine-learning).
The CFU associated with the activities of independent choice can be earned in the first or second year.
(**) “Further Knowledge” can be acquired by the student as part of the work for the preparation of the Thesis. The acquisition of such knowledge must be certified through an AC form, countersigned by the supervisor of the Degree Thesis. “Further Knowledge” can also be acquired through extramoenia or intramoenia internships. The extramoenia internship is carried out at companies, research centres or other public and/or private bodies, Italian or foreign, with the assistance of a tutor from the company or institution and the supervision of a university tutor. The intramoenia internship is carried out at university research laboratories with the assistance of a university tutor (teacher or researcher).
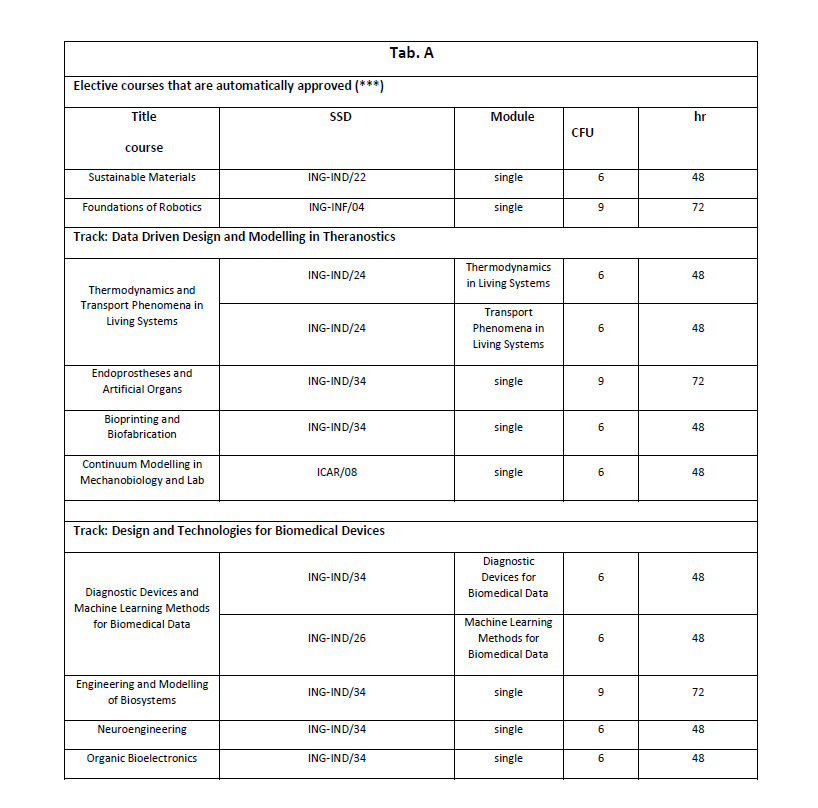
(***) For students who choose the “Data Driven Design and Modelling in Theranostics” track, all courses in the “Design and Technologies for Biomedical Devices” track are considered automatically approved.
For students who choose the “Design and Technologies for Biomedical Devices” track, all courses in the “Data Driven Design and Modelling in Theranostics” track are considered automatically approved.
List of propaedeuticities
Eligibility and Admission Guidelines
Curricular requirements: students with a BSc degree in Information Engineering (class L-8) or a degree in Industrial Engineering (class L-9) may enroll in the course.
Personal preparation requirements: Students must have knowledge of mathematical analysis, physics and electromagnetism is required. And a basic knowledge of mechanics, thermodynamics and transport phenomena is strongly advised. Students who have achieved a weighted average grade of 24.00/30.00 in the first level degree program are automatically exempted from the personal preparation assessment.
A B2 level (CEFR) English language certification is required for all students
Requests for enrollment from foreign students are evaluated on a case-by-case basis. As a general rule, students with a degree in Chemical Engineering, Materials Engineering, Biomedical Engineering, and Electronic Engineering are admitted to the assessment.


